Analysis of ICGN mice
Summary of ICGN mice
ICR-derived glomerulonephritis (ICGN) mice, a good experimental model for human idiopathic nephrotic syndrome, represent the effacement of podocyte foot processes and the thickened glomerular basement membrane (GBM) soon after birth. The ICGN mice develop proteinuria, hypoproteinemia and hyperlipidemia, and severe systemic edema. Recently, we found that the tensin2 gene of the ICGN mouse possessed an 8-nucleotide deletion mutation in exon 18, leading to a frameshift and giving rise to a terminal codon at a premature position [Refs.1 and 2].
Mutation of Tensin2
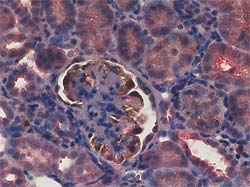
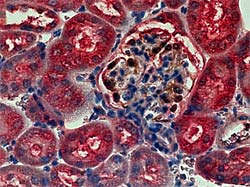
We found that the tensin2 gene showed a mutation in the ICGN mouse. The tensin2 cDNA extracted from the ICGN mouse kidney revealed a deletion of 8 nucleotides (nt1477-nt1484) in exon 18 of the ICGN tensin2 gene [ref.1]. The immunohistochemical double-staining revealed that positive signals for tensin2 were detected in podocytes in the glomeruli and the tubular epithelial cells of the ICR but not of the ICGN mouse kidney (see figures below).
| ICGN | ICR |
|---|---|
|
|
Figures
Kidney sections from 3.5-month-old ICR (left) and ICGN (right) mice. For immunohistochemistry with double staining, sections were first incubated with anti-Wilms’ tumor antigen-1, a marker for the podocytes, Envision-horseradish peroxidase, and treated with diaminobenzidine. Then, the sections were incubated with anti-tensin2, Envision-Alkaline Phosphatase, and New Fuchsin.
Histological analyses
Histopathological analyses demonstrated glomerular lesions consisting of thickened GBM and enlargement of the mesangial area without cellular proliferation.
In glomeruli and tubulointerstitium of ICGN mice as compared with ICR mice, which were used as healthy normal control, the excessive accumulation of extracellular matrix (ECM) components, type I and III collagens, and fibronectin, was observed [Ref.3]. We compared the activities of matrix metalloproteinases (MMPs), which are responsible for ECM degradation, in the kidneys of ICGN and control ICR mice [Ref.4]. We reported that lower activities of MMP-1 and MMP-2 and -9 were demonstrated in the kidneys of ICGN mice as compared with those of ICR mice, but there was no significant difference in the MMP-3 activities between these strains [Ref. 4], indicating decreased MMP activities cause abnormal accumulation of ECM in ICGN mouse kidneys. Furthermore, we demonstrated TGF-β1 induces such increases in ECM components [Ref.5].
References
- Cho A-R, Uchio-Yamada K, Torigai T, Miyamoto T, Miyoshi I, Matsuda J, Kurosawa T, Kon Y, Asano A, Sasaki N, Agui T. Deficiency of the tensin2 gene in the ICGN mouse, an animal model for congenital nephrotic syndrome. Mammalian Genome, 2006 May;17(5):407-416
- Chen H, Duncan IC, Bozorgchami H, Lo SH. Tensin1 and a previously undocumented family member, tensin2, positively regulate cell migration. Proc Natl Acad Sci U S A. 2002 Jan 22; 99(2):733-738.
- Uchio K, Manabe N, Kinoshita A, Tamura K, Miyamoto M, Ogura A, Yamamoto Y, Miyamoto H, Abnormalities of extracellular matrices and transforming growth factor beta1 localization in the kidney of the hereditary nephrotic mice (ICGN strain). J Vet Med Sci.,1999, 61, 769-776
- Uchio K, Manabe N, Tamura K, Miyamoto M, Yamaguchi M, Ogura A, Yamamoto Y, Miyamoto H., Decreased matrix metalloproteinase activity in the kidneys of hereditary nephrotic mice (ICGN strain). Nephron, 2000, 86, 145-151
- Goto Y, Manabe N, Uchio-Yamada K, Yamaguchi-Yamada M, Inoue N, Yamamoto Y, Ogura A, Nagano N, Miyamoto H., Augmented cytoplasmic Smad4 induces acceleration of TGF-β1 signaling in renal tubulointerstitial cells of hereditary nephrotic ICGN mice with chronic renal fibrosis; possible role for myofibroblastic differentiation. Cell Tissue Res., 2004, 315, 209-221
